Great news!
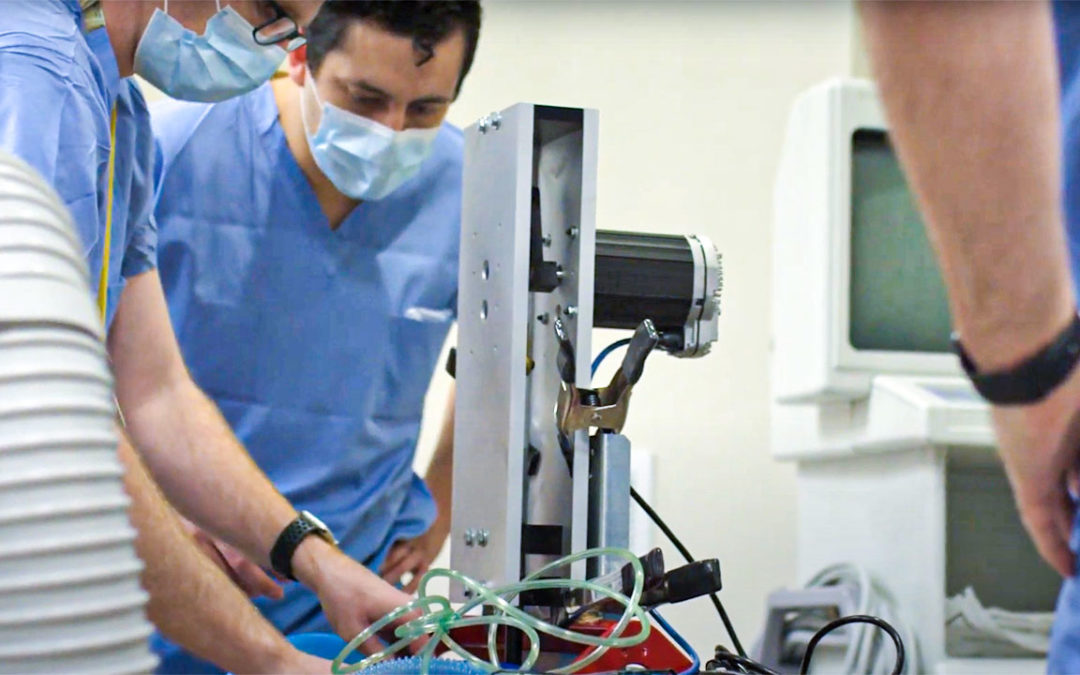
The Food and Drug Administration yesterday issued emergency use authorization (EUA) for the “Coventor” ventilator prototype developed by the Bakken Medical Devices Center at the University of Minnesota. Ajax worked directly with the project leaders at BMDC to produce enclosures for the prototype.
 The FDA’s approval enables the project to move from prototype to finished product. Ajax colleagues are now forming enclosures for the manufacturer. About 1,400 units will be completed by early next week.
The FDA’s approval enables the project to move from prototype to finished product. Ajax colleagues are now forming enclosures for the manufacturer. About 1,400 units will be completed by early next week.
Our thanks go out to all the Ajax colleagues who have been putting in long hours to form and deliver parts for this emergency life-saving machine.
Some background on the project is here.
Press on, Ajax!